












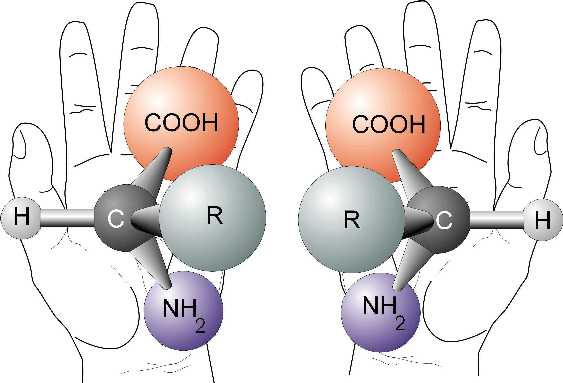
The term chiral (pronounced /ˈkaɪrəl/) is used to describe an object that is non-superposable on its mirror image.
Human hands are perhaps the most universally recognized example of chirality: The left hand is a non-superposable mirror image of the right hand; no matter how the two hands are oriented, it is impossible for all the major features of both hands to coincide. This difference in symmetry becomes obvious if someone attempts to shake the right hand of a person using his left hand, or if a left-handed glove is placed on a right hand. The term chirality is derived from the Greek word for hand, χειρ (cheir). It is a mathematical approach to the concept of "handedness".
When used in
the context of chemistry, chirality usually refers to molecules. Two
mirror images of a molecule that cannot be superposed onto each other
are referred to as enantiomers or optical isomers.
Because the difference between right and left hands is universally
known and easy to observe, many pairs of enantiomers are designated as "right-"
and "left-handed."
A mixture of equal amounts of the two enantiomers is said to be a racemic mixture. Racemic mixtures
are not optically active even though their two enantiomer constituents
when separated are. Molecular chirality is of interest because of its
application to stereochemistry in inorganic
chemistry, organic
chemistry, physical
chemistry, biochemistry,
and supramolecular
chemistry.
Many biologically active molecules are chiral, including the naturally occurring amino acids (the building blocks of proteins), and sugars. In biological systems, most of these compounds are of the same chirality: most amino acids are L and sugars are D. Typical naturally occurring proteins, made of L amino acids, are known as left-handed proteins, whereas D amino acids produce right-handed protein
Enzymes, which are chiral, often distinguish between the two enantiomers of a chiral substrate. Imagine an enzyme as having a glove-like cavity that binds a substrate. If this glove is right-handed, then one enantiomer will fit inside and be bound, whereas the other enantiomer will have a poor fit and is unlikely to bind.
D-form amino acids tend to taste sweet, whereas L-forms are usually tasteless. Spearmint leaves and caraway seeds, respectively, contain L-carvone and D-carvone - enantiomers of carvone. These smell different to most people because our olfactory receptors also contain chiral molecules that behave differently in the presence of different enantiomers.
Source: http://en.wikipedia.org/wiki/Chirality_(chemistry)